About our project

A new way to take peptide medicine – through a film inside the cheek
Current challenges
Why we need a better way
- Few peptide-therapies have been turned into tablets or capsules. However, swallowing them as tablets/capsules often does not work well, because our stomach and gut break them down, causing problems dosage control and how much of the drug actually reaches the bloodstream to do its job. They also are not absorbed well through the gut wall, and interact with food and drink.
- Injections work, but they can be painful, inconvenient, and discouraging to take regularly.

Our solution
The BUCCAL-PEP film
To overcome these challenges in oral delivery, including interactions with food and drink or being broken down by the liver, the BUCCAL-PEP consortium is developing a multifunctional, thin film that sticks to the inside of the cheek. This allows – for the first time – delivery of peptide-analogue therapies through the cheek, so-called buccal delivery. Through unique integration of an absorption booster with the peptide medicine, the film enables peptides to diffuse across the cheek.

Understanding the film
How will the BUCCAL-PEP film work?
The BUCCAL-PEP film will be placed inside the cheek and work in three steps:
1
The sticky layer releases an absorption booster that helps the cheek absorb peptides.
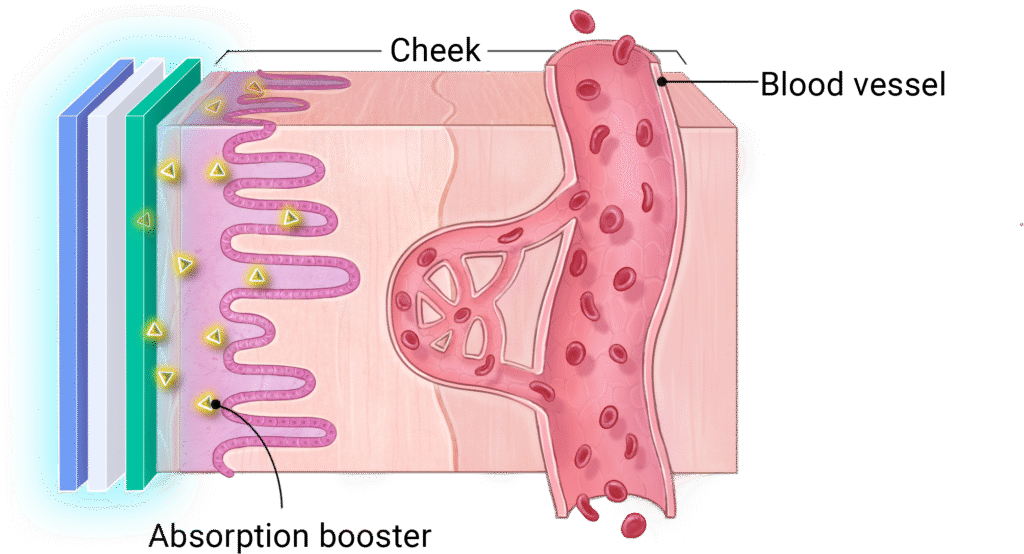
2
Small proteins (peptides) can now travel across the cheek surface to reach the blood.
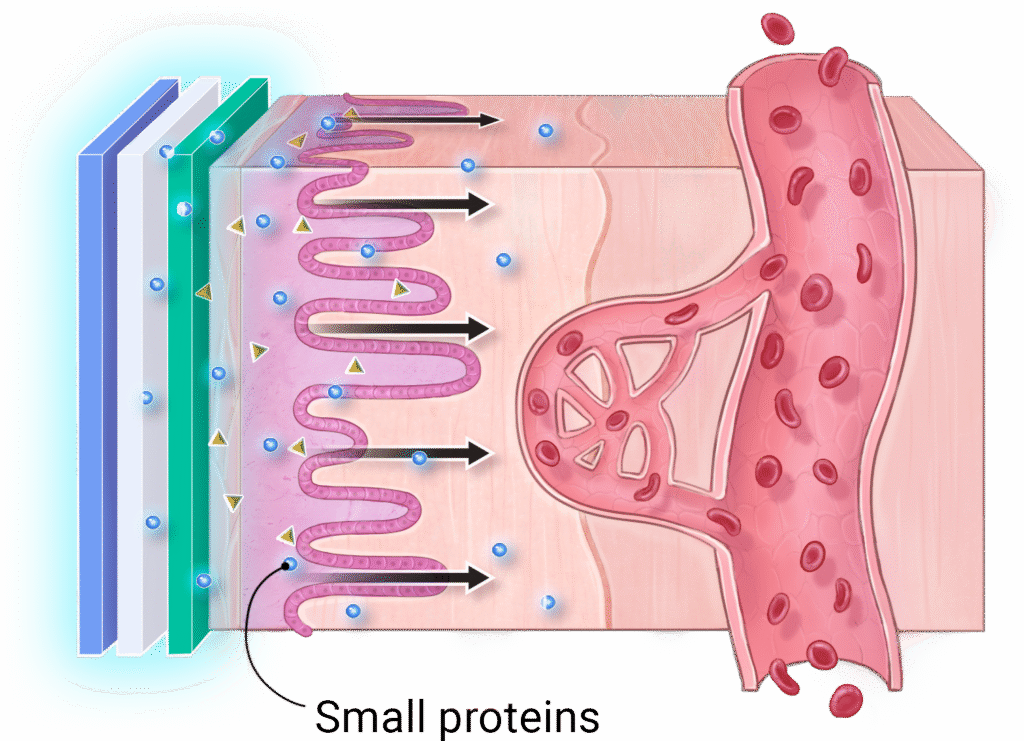
3
The film dissolves after the proteins are taken up into the blood stream.
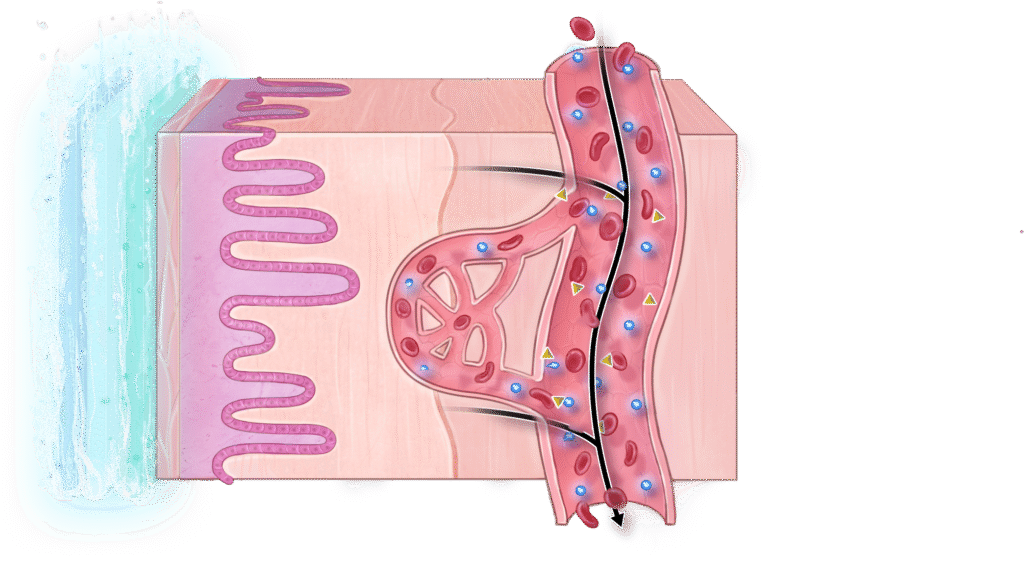
By the end of the project, we aim to have a ready-to-test film that can move into clinical trials. This could mark the start of a new generation of peptide therapies that are safer, easier, and better for patients.
Our work
How is the project structured?
In this project, the consortium will design, select and manufacture a lead product, with Type 2 Diabetes as showcase indication. The film will be validated for performance in in vivo large animal studies. Additionally, a Health Technology Assessment will be performed to support the development of an evidence-based value proposition as well as a commercialisation strategy. The final deliverable of the project is a film that is pre-clinically validated and ready for clinical trials in the desired showcase indication.
The work has been structured in different work packages:
This work package aims to develop a film that can deliver semaglutide through the cheek by optimizing the combination of different materials and ratios. The performance of different prototypes will be assessed in vitro, ex vivo, and in vivo in pig models before selecting a candidate film for further testing.
The work package will perform in vivo absorption tests on two lead film prototypes developed in WP1 in pigs. They will analyse data from blood samples collected during intravenous administration and buccal absorption tests, and provide feedback to WP3. The study will also evaluate the safety using histological analysis of the cheek surface in cross-over pig studies.
This work package’s main objective is to develop a business plan for the proposed technology platform by conducting a health economics and market assessment. This includes analysing the target population segment, assessing the commercial viability and return of investment compared to the standard of care. WP4 will also develop a comprehensive business plan for the first target indication of the BUCCAL-PEP film, while also exploring the technology’s potential for other indications of interest.
The objective of this work package is to ensure effective communication and dissemination of the project’s developments to stakeholders, including scientific community, industry, clinical experts, patients, and caregivers. The goals are to engage all stakeholders, consider their needs, communicate project progress and results to the general public, and develop a solid foundation for the exploitation strategy of the BUCCAL-PEP film.
The main objective of work package 6 is to ensure productive project execution and realisation of its main objectives in time and within budget. It also ensures performance of activities by project partners and reporting according to EC regulations.











